Preliminary results from clinical trials of an experimental antiviral drug for coronavirus could come in a week, a top researcher said Sunday.
The final test results for the drug, remdesivir, aren’t expected until mid-to-late May, said Dr. Andre Kalil, a principal investigator for the trial. But he said the team might “potentially have some early data in the next one or two weeks.”
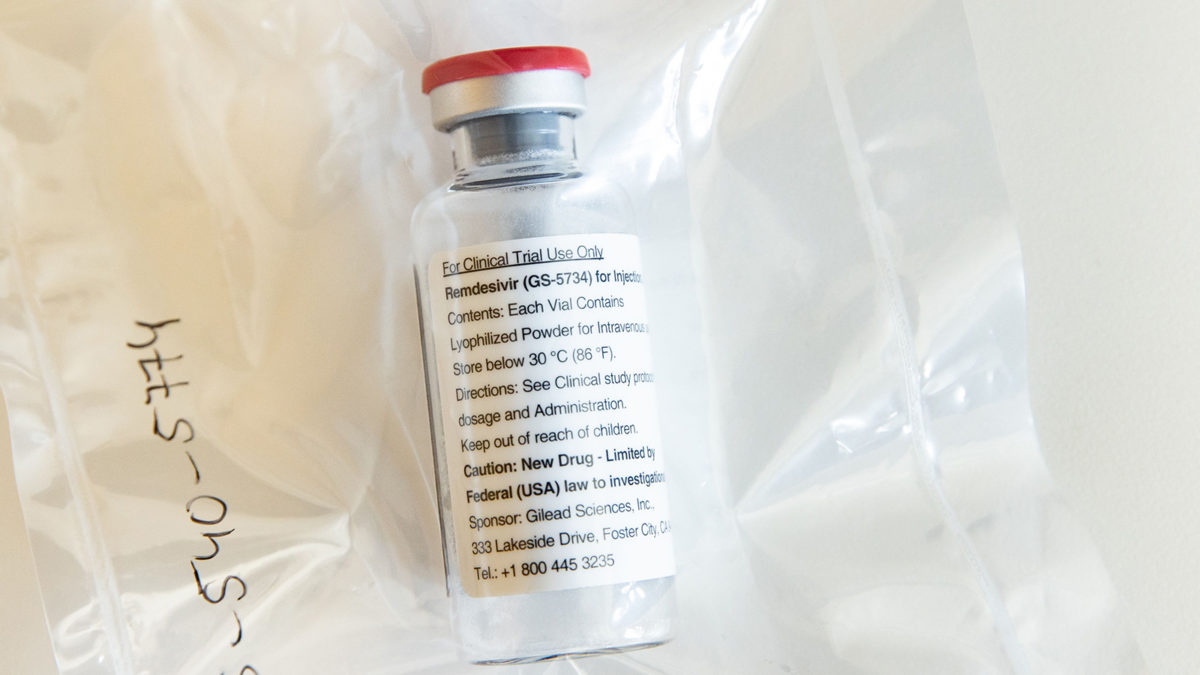
Remdesivir was originally tested by Gilead Sciences as a potential treatment for Ebola, and it showed activity against the novel coronavirus in test tubes. But whether the drug is an effective treatment for Covid-19 remains unclear.
The new study, sponsored by the National Institutes of Health, is a randomized, double-blind, placebo-controlled trial — meaning neither the patients nor their doctors know who is receiving the real drug and who is receiving a placebo.
Patients finished enrolling for the study last Sunday, Kalil said, adding that their number had exceeded the target of 572.
The trial began at the University of Nebraska Medical Center, where Kalil is a professor of medicine, but it has expanded to nearly 70 sites around the world, from South Korea to Germany.
Data on remdesivir is confusing at best. Earlier this month, the maker of the drug, Gilead released information on 53 patients, most of whom showed improvement after receiving infusions of remdesivir.
Information leaked to STAT News suggested that patients receiving remdesivir were recovering quickly, but the report was based on a recorded discussion of a clinical trial, and offered few details.
Last week, the World Health Organization accidentally published a summary of results from a trial of the drug in coronavirus patients. A screenshot published by STAT showed “remdesivir use was not associated with a difference in time to clinical improvement.”
But that trial was terminated early due to low enrolment, and Gilead said it was inconclusive.